AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
  
There are three molecular geometries: Octahedral Square pyramidal Square planar There are three molecular geometries: tetrahedral, if all are bonding pairs, trigonal pyramidal, if one is a nonbonding pair, and bent, if there are two nonbonding pairs.Īll positions are equivalent in the octahedral domain. There are two distinct positions in this geometry: Axial Equatorial Lone pairs occupy equatorial positions. There are four distinct molecular geometries in this domain: Trigonal bipyramidal Seesaw T-shaped Linear There are two molecular geometries: trigonal planar, if all electron domains are bonding, and bent, if one of the domains is a nonbonding pair. NOTE: If there are only two atoms in the molecule, the molecule will be linear no matter what the electron domain is. In the linear domain, there is only one molecular geometry: linear. 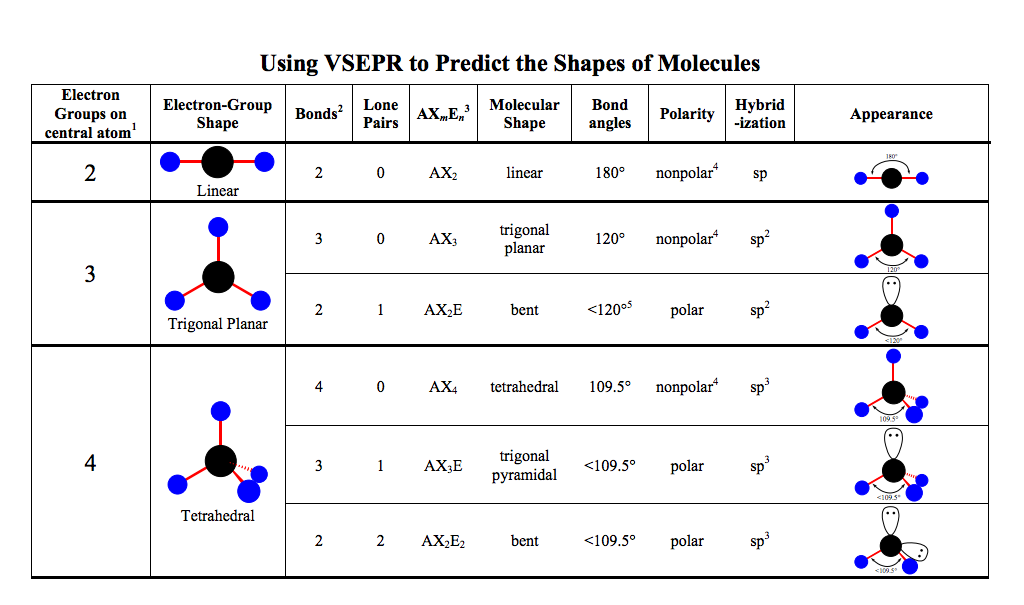
We will look at each electron domain to see what molecular geometries are possible. Tables 9.2 and 9.3 show the potential molecular geometries. To determine the electron-domain geometry, count the total number of lone pairs, single, double, and triple bonds on the central atom.ġ1 Molecular Geometries Once you have determined the electron-domain geometry, use the arrangement of the bonded atoms to determine the molecular geometry. The Table shows the electron-domain geometries for two through six electron domains around a central atom. “The best arrangement of a given number of electron domains is the one that minimizes the repulsions among them.” (The balloon analogy in the figure to the left demonstrates the maximum distances, which minimize repulsions.) The central atom in this molecule, A, has four electron domains.ĩ Valence-Shell Electron-Pair Repulsion (VSEPR) Model This is true whether there is one or more electron pairs pointing in that direction. This is the Valence-Shell Electron-Pair Repulsion (VSEPR) model.Ĩ Electron Domains We can refer to the directions to which electrons point as electron domains. By assuming the electron pairs are placed as far as possible from each other, we can predict the shape of the molecule. Simply put, electron pairs, whether they be bonding or nonbonding, repel each other. Here we see some common shapes for molecules with two or three atoms connected to a central atom.ħ What Determines the Shape of a Molecule? However, we use Lewis Structures to help us determine shapes. Kirby Quinnipiac University Hamden, CTĦ Molecular Shapes Lewis Structures show bonding and lone pairs, but do not denote shape. Lecture Presentation Chapter 9 Molecular Geometry and Bonding Theories James F. Presentation on theme: "Chapter 9 Molecular Geometry and Bonding Theories"- Presentation transcript:ġ Chapter 9 Molecular Geometry and Bonding Theories 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
